
CH1211
EDTA Disodium salt dihydrate Lab
$37.62 – $559.62
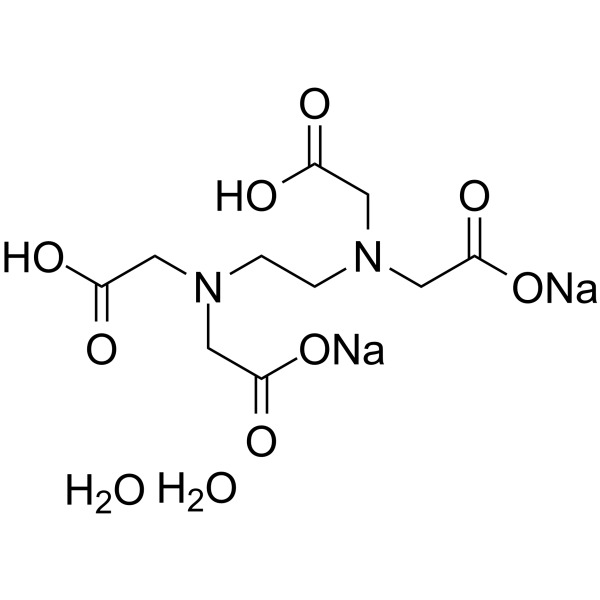
EDTA Disodium salt dihydrate Lab Grade CAS: 6381-92-6 | M.F: C10H14N2Na2O8•2H2O | MW: 372.24, Buy now for reliable and reproducible results from ChemieR Organic Reagent.
EDTA Disodium Salt Dihydrate Lab Grade (CAS: 6381-92-6) is the disodium salt form of edetate and is presented as a white or white crystalline powder. Ethylenediaminetetraacetic acid (EDTA) is an amino polycarboxylic acid that is also a hexadentate ligand and has great chelating abilities. It has a chemical formula C10H14N2Na2O8•2H2O and a molecular weight of 372.24. It is also referred to as Disodium Ethylenediaminetetraacetate Dihydrate, Disodium Ethylenediaminetetraacetate Salt Dihydrate, EDTA Disodium Salt, EDTA-Na2, Edathamil, Edetate Disodium Salt Dihydrate, and Sequestrene Na2. EDTA is a good chelator of divalent cations. This reagent is commonly used in laboratories and is able to support numerous scientific, analytical and biochemical procedures.
Applications
- Biochemical Research: EDTA Disodium Salt Dihydrate Lab Grade is also used in the making of buffer solutions that are necessary in the study of biochemical and molecular biology.
- Laboratory Applications: Complexometric titrations are used in determining the concentration of certain metal ions in solution, so that when laboratory tests are done, the measurements are precise.
Key features
- Laboratory Chemical of Economy: EDTA Disodium Salt Dihydrate Lab Grade is both economical and high performing, which is why it will be ideal in the research laboratories that have limited financial resources.
- General Purpose Scientific Reagent: The compound has a wide range of applications in biochemical investigation, in the field of analytical chemistry and in routine laboratory work.
- Safety and Ease of Handling: The EDTA Disodium Salt Dihydrate Lab Grade(CAS: 6381-92-6) is relatively easy and harmless to operate and reduces the operational hazards in the lab settings.
General Description
EDTA Disodium Salt Dihydrate Lab Grade (CAS: 6381-92-6), also referred to as the disodium salt of edetate, is a fundamental laboratory chemical supplied as a white or almost white crystalline powder. It has a molecular weight of 372.24 and molecular formula C10H14N2Na2O8·2H2O and is very common as a chelating agent used in scientific studies. This compound is an amino polycarboxylic acid and hexadentate ligand derived by Ethylenediaminetetraacetic acid (EDTA), and is appreciated due to its high binding capacity with metal ions. It has a high chelatability with divalent cations, so it is of great importance in the application of molecular biology, biochemistry, and in the analysis of chemistry.
Would you like to buy EDTA Disodium Salt Dihydrate Lab Grade (CAS: 6381-92-6)? ChemieR Organic Reagents offers EDTA Disodium Salt Dihydrate of laboratory-grade that is capable of providing reliable and reproducible results in both research and testing processes. The reason behind its regular purity and reliable performance is that it can be used in regular laboratory and research-driven settings. Customers who want to have competitive prices, bulk buying possibilities, and reliability in the suppliers of the laboratory chemical products may order EDTA Disodium Salt Dihydrate Lab Grade in order to obtain precision, accuracy and quality-based scientific results. Order EDTA Disodium Salt Dihydrate Lab now to guarantee precision, consistency, and reliable outcomes in your lab and industrial usage.
About Brand
ChemieR provides an extensive selection of high-purity salts, acids, base and high purity solvents, including alkali metals, alkaline earth compounds, perchlorates, heavy metals, rare earths, transition metals, silver and precious metals, and triflates. ChemieR specialize in inorganic chemicals, offering materials with low moisture content and minimal trace metals. ChemieR stands out with a diverse range of laboratory chemicals, from organic and inorganic reagents to solvents.
Related Products
Redirect Notice
ChemieR products are exclusively distributed by Dawn Scientific.
You are now being redirected to www.dawnscientific.com to complete your purchase.
Thank you.










