
Sodium Bicarbonate ACS Grade CAS: 144-55-8 | M.F: NaHCO3 | MW: 84.01, Buy now for reliable and Reproducible results from ChemieR Inorganic Reagent.
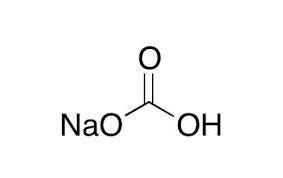
Sodium Bicarbonate ACS Grade (CAS No. 144-55-8), also known as Baking Soda, is a white crystalline solid typically found in fine powder form. It has moderate saline and alkali flavor that reminds of washing soda (sodium carbonate). The compound has the chemical formula NaHCO3 and consists of sodium ions (Na+) and bicarbonate ions (HCO3-). Sodium bicarbonate dissolves very well in water, hence it has many applications in the laboratory and industry. ACS grade quality will guarantee high purity, which is vital in the acquisition of accurate and repeatable results in scientific tests and analytical processes.
Applications
- Sodium Bicarbonate ACS Grade (CAS No. 144-55-8) is commonly utilized in titrations of laboratories, biochemical analysis, the preparation of buffer solutions, and the control of pH.
- It is common in the biological and chemical buffer systems where it assists in stabilizing a pH environment during the reaction of chemicals.
- In co-precipitation techniques, Sodium Bicarbonate ACS acts as a precipitating agent in the synthesis of gamma alumina (γ-Al2O3) catalysts, which are important catalysts used in methanol dehydration reactions for producing dimethyl ether.
Key features
- Sodium bicarbonate ACS is of high purity to the standard of ACS to guarantee dependable chemical purity and laboratory and analytical use.
- The product possesses good chemical stability and thus is effective and safe to conduct laboratory tests and use in industries.
- Sodium Bicarbonate ACS is a low-cost reagent that could be used in the labs, research facilities, and manufacturing plants.
- The compound is very easy to use, and is safe in normal laboratory practices, and is environmentally friendly.
General Description
Sodium Bicarbonate ACS (CAS No. 144-55-8), commonly referred to as Baking Soda, is a white crystalline salt-like material in the form of a slightly alkaline and slightly soapy flavor that resembles that of washing soda. This compound consists of sodium ions and bicarbonate ions and its molecular formula is NaHCO3. Due to its great solubility and versatility in chemistry, Sodium Bicarbonate ACS is highly applicable in laboratories, research and other industries. The ACS-grade purity of Sodium Bicarbonate also guarantees that it is of high standards needed in laboratory experiments, analytical chemistry and scientific research which enables researchers to get precise and repeatable results.
In case laboratories and industries want to find Sodium Bicarbonate ACS Grade (CAS No. 144-55-8), then ChemieR Inorganic Reagents offer quality laboratory chemicals that are expected to perform reliably. They find extensive application in scientific experiments, industrial applications, and research settings where reliable quality of chemicals is required. Sodium Bicarbonate ACS can have a price change according to the quantity and packaging features. The site or customer service can provide detailed information to customers who want to know the latest pricing and availability of the products. The competitive price alternatives also enable the labs and industries to access quality chemical reagents at reduced costs. Order online Sodium Bicarbonate ACS Grade (CAS No. 144-55-8) from ChemieR today and receive your products fast, secure, and reliably so that you succeed in your project.
About Brand
ChemieR provides an extensive selection of high-purity salts, acids, base and high purity solvents, including alkali metals, alkaline earth compounds, perchlorates, heavy metals, rare earths, transition metals, silver and precious metals, and triflates. ChemieR specialize in inorganic chemicals, offering materials with low moisture content and minimal trace metals. ChemieR stands out with a diverse range of laboratory chemicals, from organic and inorganic reagents to solvents.
Related Products
Redirect Notice
ChemieR products are exclusively distributed by Dawn Scientific.
You are now being redirected to www.dawnscientific.com to complete your purchase.
Thank you.










